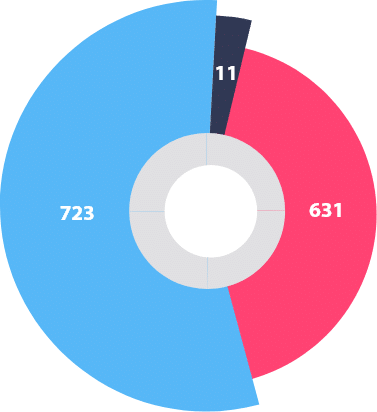
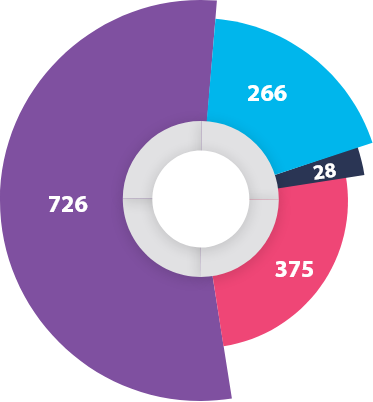
R&D Strength
Suven Pharma has an in house team of highly qualified professionals to ensure customer satisfaction.
Suven Pharma provides state-of-the-art R&D facilities to handle challenging product development. Our core competency is the rapid transfer of technology from the lab to the commercialization level. We flourish in the development of new product processes in association with top global pharmaceutical and fine chemical companies.